-
已知在25°C、101 kPa下,1 g C8H18(辛烷)燃烧生成CO2和液态水时放热48.40 kJ。表示上述反应的热化学方程式正确的是 ( )
A.C8H18(l)+25/2O2(g)=8CO2(g)+9H2O(g) ΔH=-5518 kJ/mol
B.C8H18(l)+25/2O2(g)=8CO2(g)+9H2O(l) ΔH=-5518 kJ/mol
C.C8H18(l)+25/2O2(g)=8CO2(g)+9H2O(l) ΔH=+5518 kJ/mol
D.C8H18(l)+25/2O2(g)=8CO2(g)+9H2O(l) ΔH=-48.40 kJ/mol
高一化学选择题中等难度题查看答案及解析
-
已知在25℃,101KPa下,1g C8H18(辛烷)燃烧生成二氧化碳和液态水时放出48.40KJ热量。表示上述反应的热化学方程式正确的是
A.C8H18(l)+12.5O2(g)== 8CO2(g) + 9H2O(g) ΔH= ‒ 48.40KJ/mol
B.C8H18(l)+12.5O2(g)== 8CO2(g) + 9H2O(l) ΔH= ‒ 5518KJ/mol
C.C8H18(l)+12.5O2(g)== 8CO2(g) + 9H2O(l) ΔH= +5518KJ/mol
D.C8H18(l)+12.5O2(g)== 8CO2(g) + 9H2O(l) ΔH= ‒ 48.40KJ/mol
高一化学选择题简单题查看答案及解析
-
已知在25℃,101KPa下,1g C8H18(辛烷)燃烧生成二氧化碳和液态水时放出48.40KJ热量。表示上述反应的热化学方程式正确的是
A.C8H18(l)+12.5O2(g)== 8CO2(g) + 9H2O(g) ΔH= —48.40KJ/mol
B.C8H18(l)+12.5O2(g)== 8CO2(g) + 9H2O(l) ΔH= —5518KJ/mol
C.C8H18(l)+12.5O2(g)== 8CO2(g) + 9H2O(l) ΔH= +5518KJ/mol
D.C8H18(l)+12.5O2(g)== 8CO2(g) + 9H2O(l) ΔH= —48.40KJ/mol
高一化学选择题简单题查看答案及解析
-
已知在25℃,101KPa下,1g C8H18(辛烷)燃烧生成二氧化碳和液态水时放出48.40KJ热量。表示上述反应的热化学方程式正确的是( )
A、C8H18(l)+12.5O2(g)== 8CO2(g) + 9H2O(g)ΔH=-48.40KJ/mol
B、C8H18(l)+12.5O2(g)== 8CO2(g) + 9H2O(l)ΔH=-5518KJ/mol
C、C8H18(l)+12.5O2(g)== 8CO2(g) + 9H2O(l)ΔH=+5518KJ/mol
D、C8H18(l)+12.5O2(g)== 8CO2(g) + 9H2O(l)ΔH=-48.40KJ/mol
高一化学选择题简单题查看答案及解析
-
已知在25℃,101kPa下,lgC8H18(辛烷)燃烧生成二氧化碳和液态水时放出48.40kJ热量。表示上述反应的热化学方程式正确的是
A.C8H18(1)+12.5O2(g)=8CO2(g)+9H2O(g);△H=-48.40kJ·mol-1
B.C8H18(1)+12.5O2(g)=8CO2(g)+9H2O(1);△H=-5518kJ·mol-1
C.C8H18(1)+12.5O2(g)=8CO2(g)+9H2O(1);△H=+5518kJ·mol-1
D.C8H18(1)+12.5O2(g)=8CO2(g)+9H2O(1);△H=-48.40kJ·mol
高一化学选择题简单题查看答案及解析
-
已知在25℃,101kPa下,1g辛烷C8H18燃烧生成二氧化碳和液态水时放出48.40kJ热量.表示上述反应的热化学方程式正确的是 ( )
A.
△H=-48.40kJ/mol
B.
△H=-5518kJ/mol
C.
△H=+5518kJ/mol
D.
△H=+11036kJ/mol
高一化学选择题简单题查看答案及解析
-
已知在25 ℃、101 kPa下,1 g C8H18(l)燃烧生成CO2和液态H2O时放出48.40 kJ的热量,表示上述反应的热化学方程式正确的是
A.C8H18(l)+
O2(g)===8CO2(g)+9H2O(g) ΔH=-48.40 kJ·mol-1
B.C8H18(l)+
O2(g)===8CO2(g)+9H2O(l) ΔH=-5 517.6 kJ·mol-1
C.C8H18(l)+
O2(g)===8CO2(g)+9H2O(g) ΔH=+5 517.6 kJ·mol-1
D.2C8H18(l)+25O2(g)===16CO2(g)+18 H2O(l) ΔH=-5 517.6 kJ·mol-1
高一化学选择题中等难度题查看答案及解析
-
已知在25℃,101kPa下,57gC8H18(辛烷)燃烧生成二氧化碳和液态水时放出2758.80kJ热量。表示上述反应的热化学方程式正确的是
A.C8H18(l)+12.5O2(g)=8CO2(g)+9H2O(g)△H=-48.40kJ·mol-1
B.C8H18(1)+12.5O2(g)=8CO2(g)+9H2O(1)△H=-5517.6kJ·mol-1
C.C8H18(1)+12.5O2(g)=8CO2(g)+9H2O(1)△H=+5517.6kJ·mol-1
D.C8H18(1)+12.5O2(g)=8CO2(g)+9H2O(1)△H=-48.40kJ·mol-1
高一化学选择题中等难度题查看答案及解析
-
已知在25℃,101kPa下,1gC8H18(辛烷)燃烧生成二氧化碳和液态水时放出48.40kJ热量.表示上述反应的热化学方程式正确的是( )
A.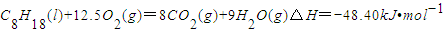
B.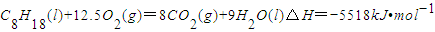
C.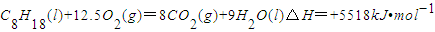
D.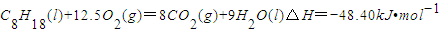
高一化学选择题中等难度题查看答案及解析
-
已知在25 ℃、101 kPa 下,11.4 g C8H18(辛烷)燃烧生成二氧化碳和液态水时放出 551.76 kJ 能量。表示上述反应的热化学方程式正确的是( )
A.C8H18(l)+
O2(g)=8CO2(g)+9H2O(g) ΔH=-551.76 kJ·mol-1
B.C8H18(l)+
O2(g)=8CO2(g)+9H2O(l) ΔH=-5517.6 kJ·mol-1
C.C8H18(l)+
O2(g)=8CO2(g)+9H2O(l) ΔH=5517.6 kJ·mol-1
D.C8H18(l)+
O2(g)=8CO2(g)+9H2O(l) ΔH=-551.76 kJ·mol-1
高一化学单选题简单题查看答案及解析